Noble Gas Notation For Arsenic
QUESTION 10 A arsenic atom needs to gain A ii electrons to accomplish noble gas configuration B. vi C 4 D.3 €. 5

Related Question
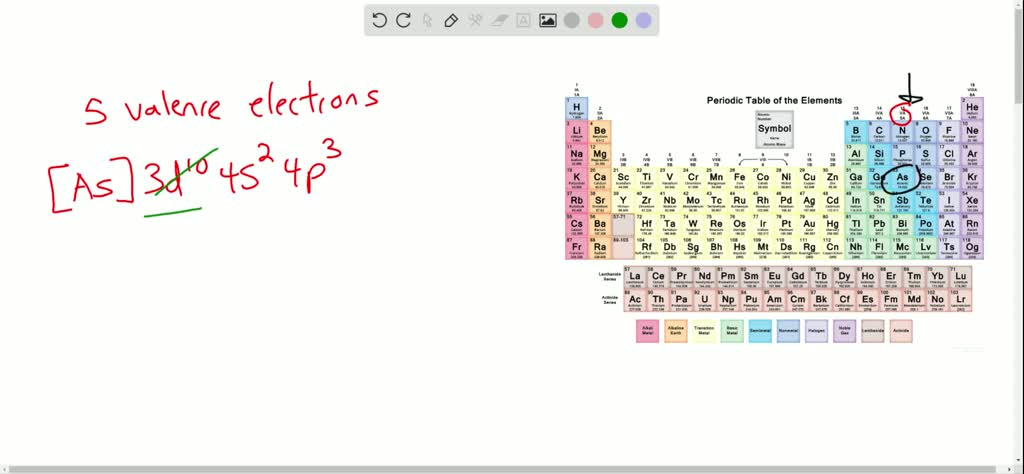
The electron configuration for arsenic, Every bit, is $[\mathrm{Ar}] 3 d^{10} 4 s^{2} four p^{3} .$ How many valence electrons does an Every bit cantlet have? Write the symbol for the ion it forms to attain a noble-gas configuration.
Discussion
Y'all must be signed in to hash out.
Video Transcript
All correct, this question. Nosotros're gonna be talking nigh arsenic. And then arsenic is over here on the periodic table of the aforementioned grouping as nitrogen. It's in grouping 5 a specifically. So that means it has 5 valence electrons. Only let's say you lot didn't have announced out of table handy, and all that yous were given was the configuration. And so it would be three d ton for united states to and before p three. And so we know that in the configuration, that D orbital is a lower free energy or bill and information technology's in the tertiary show. So we're going Teoh, ignore the electrons in that d orbital when nosotros're talking about the valence electrons, then I only have two Dio is out of the to transfer the s orbital in the three electrons from the PR abound in that equals five residual similar chance. So how exercise we get arsenic to have a total viii surveillance electrons for the octet rule? Well, if we draw our arsenic and they used the term depict out very loosely, we dropped the lewis dot structure for the states make. Nosotros have arsenic with 5 electrons. Each of these dots represents an electron. So to get a total pay. We have to add 3 more electrons. Okay, so if we add together three more electrons without adding whatever more protons, we know that we change the charge to a negative 3. Then that will be written every bit arsenic. Uh, three minus. And it'south a
Noble Gas Notation For Arsenic,
Source: https://www.numerade.com/ask/question/question-10-a-arsenic-atom-needs-to-gain-a-2-electrons-to-achieve-noble-gas-configuration-b-6-c-4-d3-5-29352/
Posted by: loefflergivy1941.blogspot.com




0 Response to "Noble Gas Notation For Arsenic"
Post a Comment