Allowed Set Of Quantum Numbers
Quantum Numbers and Electron Configurations
Quantum Numbers
The Bohr model was a one-dimensional model that used one quantum number to describe the distribution of electrons in the atom. The just information that was important was the size of the orbit, which was described by the northward breakthrough number. Schr�dinger's model allowed the electron to occupy three-dimensional space. It therefore required three coordinates, or three quantum numbers, to depict the orbitals in which electrons tin can exist institute.
The iii coordinates that come from Schr�dinger's wave equations are the main (n), athwart (50), and magnetic (yard) breakthrough numbers. These quantum numbers depict the size, shape, and orientation in space of the orbitals on an atom.
The chief quantum number (due north) describes the size of the orbital. Orbitals for which northward = ii are larger than those for which n = 1, for example. Because they accept contrary electric charges, electrons are attracted to the nucleus of the atom. Free energy must therefore be absorbed to excite an electron from an orbital in which the electron is close to the nucleus (n = 1) into an orbital in which it is further from the nucleus (due north = two). The principal quantum number therefore indirectly describes the energy of an orbital.
The athwart quantum number (l) describes the shape of the orbital. Orbitals have shapes that are all-time described every bit spherical (fifty = 0), polar (l = 1), or cloverleaf (l = two). They tin even take on more complex shapes as the value of the angular quantum number becomes larger.

There is only one way in which a sphere (l = 0) can be oriented in infinite. Orbitals that have polar (fifty = 1) or cloverleaf (l = ii) shapes, however, can point in different directions. We therefore need a third quantum number, known as the magnetic quantum number (1000), to describe the orientation in space of a particular orbital. (Information technology is called the magnetic quantum number because the consequence of dissimilar orientations of orbitals was first observed in the presence of a magnetic field.)
![]()
Rules Governing the Immune Combinations of Quantum Numbers
- The three quantum numbers (north, l, and m) that describe an orbital are integers: 0, one, 2, three, and so on.
- The principal quantum number (n) cannot be zero. The immune values of n are therefore 1, 2, three, 4, and then on.
- The athwart quantum number (l) tin exist any integer between 0 and n - 1. If n = iii, for case, l tin can be either 0, one, or 2.
- The magnetic quantum number (k) can be whatsoever integer between -l and +fifty. If fifty = 2, chiliad can be either -two, -ane, 0, +1, or +2.
![]()
Shells and Subshells of Orbitals
Orbitals that have the same value of the main breakthrough number class a shell. Orbitals within a crush are divided into subshells that have the same value of the angular quantum number. Chemists describe the shell and subshell in which an orbital belongs with a two-character lawmaking such as twop or 4f. The get-go character indicates the shell (n = ii or n = 4). The second grapheme identifies the subshell. Past convention, the following lowercase letters are used to indicate dissimilar subshells.
| southward: | l = 0 | |
| p: | fifty = 1 | |
| d: | l = 2 | |
| f: | l = 3 |
Although there is no pattern in the kickoff iv letters (s, p, d, f), the letters progress alphabetically from that indicate (thou, h, and and then on). Some of the immune combinations of the due north and l breakthrough numbers are shown in the figure beneath.

The third dominion limiting allowed combinations of the n, 50, and m quantum numbers has an important consequence. It forces the number of subshells in a shell to be equal to the master breakthrough number for the crush. The n = 3 shell, for case, contains three subshells: the threes, 3p, and iiid orbitals.
![]()
Possible Combinations of Breakthrough Numbers
At that place is just one orbital in the n = one shell because there is only i way in which a sphere can be oriented in space. The only immune combination of breakthrough numbers for which n = ane is the following.
In that location are 4 orbitals in the n = two shell.
| 2 | 1 | -1 | | |||
| 2 | 1 | 0 | 2p | |||
| 2 | 1 | i |
There is only ane orbital in the 2s subshell. But, at that place are 3 orbitals in the 2p subshell because there are three directions in which a p orbital can point. One of these orbitals is oriented along the X centrality, another along the Y axis, and the tertiary along the Z axis of a coordinate organization, equally shown in the figure below. These orbitals are therefore known as the iipx , 2py , and 2pz orbitals.

At that place are ix orbitals in the n = iii crush.
In that location is one orbital in the 3s subshell and three orbitals in the 3p subshell. The due north = 3 shell, however, as well includes threed orbitals.
The five unlike orientations of orbitals in the iiid subshell are shown in the figure below. 1 of these orbitals lies in the XY airplane of an XYZ coordinate system and is called the 3d xy orbital. The iiid xz and threed yz orbitals have the same shape, only they lie betwixt the axes of the coordinate system in the XZ and YZ planes. The fourth orbital in this subshell lies along the X and Y axes and is chosen the 3dx 2 -y 2 orbital. Most of the space occupied by the 5th orbital lies along the Z axis and this orbital is chosen the threedz two orbital.
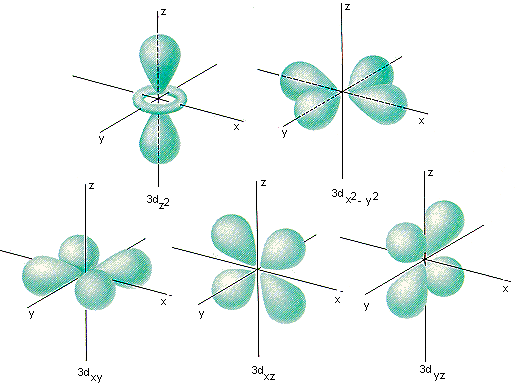
The number of orbitals in a beat is the square of the master quantum number: anetwo = i, 2two = 4, 32 = 9. There is one orbital in an s subshell (l = 0), three orbitals in a p subshell (fifty = 1), and five orbitals in a d subshell (l = ii). The number of orbitals in a subshell is therefore ii(l) + ane.
Earlier nosotros can use these orbitals we demand to know the number of electrons that can occupy an orbital and how they tin be distinguished from one another. Experimental evidence suggests that an orbital tin can agree no more than 2 electrons.
To distinguish between the 2 electrons in an orbital, we need a fourth breakthrough number. This is called the spin quantum number (s) because electrons behave every bit if they were spinning in either a clockwise or counterclockwise fashion. 1 of the electrons in an orbital is arbitrarily assigned an due south quantum number of +1/ii, the other is assigned an s quantum number of -1/2. Thus, it takes three breakthrough numbers to define an orbital but iv breakthrough numbers to identify one of the electrons that can occupy the orbital.
The allowed combinations of n, l, and m quantum numbers for the first iv shells are given in the table below. For each of these orbitals, there are ii allowed values of the spin quantum number, s.
![]()
Summary of Allowed Combinations of Quantum Numbers
| n | l | k | Subshell Notation | Number of Orbitals in the Subshell | Number of Electrons Needed to Fill Subshell | Total Number of Electrons in Subshell | |||||
| ���������������������������������������������������������������� | |||||||||||
| 1 | 0 | 0 | 1s | 1 | 2 | 2 | |||||
| ���������������������������������������������������������������� | |||||||||||
| 2 | 0 | 0 | 2s | 1 | two | ||||||
| 2 | i | one,0,-1 | 2p | three | half dozen | 8 | |||||
| ���������������������������������������������������������������� | |||||||||||
| 3 | 0 | 0 | 3s | 1 | 2 | ||||||
| three | i | 1,0,-1 | 3p | iii | 6 | ||||||
| 3 | 2 | two,i,0,-ane,-ii | 3d | 5 | x | 18 | |||||
| ���������������������������������������������������������������� | |||||||||||
| iv | 0 | 0 | 4s | i | two | ||||||
| 4 | 1 | 1,0,-one | 4p | 3 | 6 | ||||||
| four | ii | 2,1,0,-1,-2 | 4d | 5 | 10 | ||||||
| 4 | iii | 3,two,1,0,-ane,-two,-3 | 4f | 7 | 14 | 32 | |||||
![]()
The Relative Energies of Atomic Orbitals
Because of the force of attraction betwixt objects of opposite accuse, the most of import gene influencing the energy of an orbital is its size and therefore the value of the main quantum number, n. For an atom that contains but 1 electron, there is no departure betwixt the energies of the different subshells inside a shell. The 3south, 3p, and 3d orbitals, for example, accept the aforementioned energy in a hydrogen atom. The Bohr model, which specified the energies of orbits in terms of nothing more than the distance between the electron and the nucleus, therefore works for this atom.
The hydrogen cantlet is unusual, however. Equally presently as an atom contains more than than one electron, the different subshells no longer accept the same energy. Inside a given shell, the s orbitals always take the lowest free energy. The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger.
Relative energies: s < p < d < f
As a result, two factors control the energy of an orbital for about atoms: the size of the orbital and its shape, as shown in the effigy below.

A very unproblematic device can be synthetic to estimate the relative energies of atomic orbitals. The immune combinations of the northward and 50 quantum numbers are organized in a table, as shown in the figure below and arrows are drawn at 45 degree angles pointing toward the bottom left corner of the table.

The order of increasing energy of the orbitals is then read off by following these arrows, starting at the superlative of the first line and and so proceeding on to the 2d, third, fourth lines, and then on. This diagram predicts the post-obit club of increasing free energy for atomic orbitals.
1southward < twos < 2p < iiis < 3p <4s < 3d <fourp < fivesouth < fourd < fivep < 6s < 4f < fived < 6p < sevensouthward < fivef < half-dozend < 7p < viiis ...
![]()
Electron Configurations, the Aufbau Principle, Degenerate Orbitals, and Hund's Rule
The electron configuration of an cantlet describes the orbitals occupied by electrons on the cantlet. The basis of this prediction is a rule known as the aufbau principle, which assumes that electrons are added to an cantlet, ane at a fourth dimension, starting with the lowest free energy orbital, until all of the electrons have been placed in an appropriate orbital.
A hydrogen atom (Z = 1) has only one electron, which goes into the everyman energy orbital, the 1s orbital. This is indicated by writing a superscript "one" afterwards the symbol for the orbital.
H (Z = 1): 1s i
The next element has two electrons and the second electron fills the 1s orbital because in that location are only two possible values for the spin quantum number used to distinguish between the electrons in an orbital.
He (Z = 2): 1s 2
The third electron goes into the side by side orbital in the energy diagram, the twos orbital.
Li (Z = 3): 1s 2 twos 1
The fourth electron fills this orbital.
Be (Z = 4): is 2 iisouth 2
Later the 1s and 2s orbitals have been filled, the next lowest energy orbitals are the three twop orbitals. The fifth electron therefore goes into one of these orbitals.
B (Z = 5): 1s two 2s 2 2p i
When the time comes to add a sixth electron, the electron configuration is obvious.
C (Z = vi): 1southward 2 2due south 2 2p 2
However, there are three orbitals in the twop subshell. Does the 2nd electron get into the same orbital as the get-go, or does information technology go into one of the other orbitals in this subshell?
To answer this, nosotros demand to sympathize the concept of degenerate orbitals. Past definition, orbitals are degenerate when they take the same energy. The energy of an orbital depends on both its size and its shape because the electron spends more of its time farther from the nucleus of the atom as the orbital becomes larger or the shape becomes more complex. In an isolated atom, yet, the energy of an orbital doesn't depend on the direction in which it points in space. Orbitals that differ merely in their orientation in space, such equally the 2px , 2py , and 2pz orbitals, are therefore degenerate.
Electrons make full degenerate orbitals according to rules commencement stated past Friedrich Hund. Hund'south rules can be summarized equally follows.
- One electron is added to each of the degenerate orbitals in a subshell earlier two electrons are added to any orbital in the subshell.
- Electrons are added to a subshell with the aforementioned value of the spin quantum number until each orbital in the subshell has at to the lowest degree 1 electron.
When the time comes to place two electrons into the 2p subshell we put one electron into each of two of these orbitals. (The choice between the 2px , twopy , and 2pz orbitals is purely arbitrary.)
C (Z = 6): 1southward 2 2s two twopx 1 2py 1
The fact that both of the electrons in the iip subshell have the same spin quantum number can be shown by representing an electron for which south = +1/two with an
arrow pointing up and an electron for which s = -1/2 with an arrow pointing down.
The electrons in the 2p orbitals on carbon can therefore be represented as follows.
![]()
When we get to Northward (Z = 7), we have to put i electron into each of the 3 degenerate iip orbitals.
| N (Z = 7): | ones 2 2s 2 2p iii | |
Because each orbital in this subshell now contains ane electron, the next electron added to the subshell must have the reverse spin quantum number, thereby filling one of the twop orbitals.
| O (Z = eight): | 1due south 2 2s 2 twop 4 |  |
The ninth electron fills a 2d orbital in this subshell.
| F (Z = 9): | 1south 2 2s 2 twop five |  |
The tenth electron completes the 2p subshell.
| Ne (Z = ten): | 1s ii 2southward two twop 6 |  |
There is something unusually stable about atoms, such every bit He and Ne, that have electron configurations with filled shells of orbitals. By convention, nosotros therefore write abbreviated electron configurations in terms of the number of electrons across the previous element with a filled-shell electron configuration. Electron configurations of the next two elements in the periodic table, for example, could exist written as follows.
Na (Z = 11): [Ne] threes 1
Mg (Z = 12): [Ne] iiisouthward two
The aufbau procedure can be used to predict the electron configuration for an element. The actual configuration used past the element has to be determined experimentally. The experimentally determined electron configurations for the elements in the first four rows of the periodic table are given in the tabular array in the following department.
![]()
The Electron Configurations of the Elements
(1st, 2nd, 3rd, and 4th Row Elements)
| Atomic Number | Symbol | Electron Configuration | ||
| ���������������������������������������������������������������� | ||||
| 1 | H | 1s ane | ||
| two | He | 1s 2 = [He] | ||
| 3 | Li | [He] 2s i | ||
| 4 | Exist | [He] twos ii | ||
| 5 | B | [He] 2s ii 2p one | ||
| six | C | [He] 2due south 2 2p 2 | ||
| 7 | Northward | [He] twosouth 2 2p iii | ||
| 8 | O | [He] 2due south ii 2p 4 | ||
| 9 | F | [He] 2s 2 2p v | ||
| ten | Ne | [He] 2south 2 2p half dozen = [Ne] | ||
| 11 | Na | [Ne] 3s 1 | ||
| 12 | Mg | [Ne] threedue south ii | ||
| 13 | Al | [Ne] 3southward 2 iiip i | ||
| xiv | Si | [Ne] threesouthward 2 3p ii | ||
| 15 | P | [Ne] 3due south 2 3p 3 | ||
| sixteen | S | [Ne] 3s 2 3p four | ||
| 17 | Cl | [Ne] 3southward ii threep five | ||
| xviii | Ar | [Ne] 3s two 3p half dozen = [Ar] | ||
| 19 | K | [Ar] foursouthward i | ||
| 20 | Ca | [Ar] 4due south ii | ||
| 21 | Sc | [Ar] 4s 2 3d 1 | ||
| 22 | Ti | [Ar] ivs 2 iiid ii | ||
| 23 | V | [Ar] fours 2 3d iii | ||
| 24 | Cr | [Ar] ivs one 3d 5 | ||
| 25 | Mn | [Ar] 4s two 3d 5 | ||
| 26 | Atomic number 26 | [Ar] ivdue south 2 3d 6 | ||
| 27 | Co | [Ar] 4due south 2 3d 7 | ||
| 28 | Ni | [Ar] 4s two 3d 8 | ||
| 29 | Cu | [Ar] 4south one iiid 10 | ||
| 30 | Zn | [Ar] ivsouth 2 3d x | ||
| 31 | Ga | [Ar] 4s 2 threed ten ivp 1 | ||
| 32 | Ge | [Ar] 4southward 2 iiid 10 4p two | ||
| 33 | Equally | [Ar] ivs 2 threed 10 fourp 3 | ||
| 34 | Se | [Ar] fourdue south two 3d 10 4p iv | ||
| 35 | Br | [Ar] 4s 2 3d 10 ivp 5 | ||
| 36 | Kr | [Ar] fours 2 3d x 4p 6 = [Kr] | ||
![]()
Exceptions to Predicted Electron Configurations
In that location are several patterns in the electron configurations listed in the tabular array in the previous department. 1 of the most hitting is the remarkable level of understanding between these configurations and the configurations we would predict. There are but two exceptions among the first forty elements: chromium and copper.
Strict adherence to the rules of the aufbau process would predict the following electron configurations for chromium and copper.
| predicted electron configurations: | Cr (Z = 24): [Ar] 4south 2 3d 4 | |
| Cu (Z = 29): [Ar] ivs 2 3d 9 |
The experimentally determined electron configurations for these elements are slightly different.
| actual electron configurations: | Cr (Z = 24): [Ar] 4due south one 3d 5 | |
| Cu (Z = 29): [Ar] ivdue south 1 3d 10 |
In each case, one electron has been transferred from the 4due south orbital to a iiid orbital, even though the 3d orbitals are supposed to be at a higher level than the 4s orbital.
Once we get beyond atomic number twoscore, the difference betwixt the energies of adjacent orbitals is small enough that it becomes much easier to transfer an electron from 1 orbital to some other. Virtually of the exceptions to the electron configuration predicted from the aufbau diagram shown before therefore occur among elements with atomic numbers larger than 40. Although it is tempting to focus attending on the scattering of elements that have electron configurations that differ from those predicted with the aufbau diagram, the amazing affair is that this simple diagram works for and then many elements.
![]()
Electron Configurations and the Periodic Table
When electron configuration information are arranged so that we tin can compare elements in one of the horizontal rows of the periodic table, we detect that these rows typically correspond to the filling of a shell of orbitals. The second row, for example, contains elements in which the orbitals in the due north = 2 beat out are filled.
| Li (Z = 3): | [He] 2s one | |
| Be (Z = 4): | [He] twos 2 | |
| B (Z = five): | [He] twos 2 iip 1 | |
| C (Z = 6): | [He] 2s 2 iip 2 | |
| Due north (Z = 7): | [He] twos 2 2p 3 | |
| O (Z = 8): | [He] 2s 2 iip 4 | |
| F (Z = nine): | [He] 2southward 2 2p 5 | |
| Ne (Z = 10): | [He] iisouth ii 2p 6 |
There is an obvious pattern within the vertical columns, or groups, of the periodic table also. The elements in a group take similar configurations for their outermost electrons. This relationship can be seen past looking at the electron configurations of elements in columns on either side of the periodic table.
| Group IA | Group VIIA | |||||
| H | 1s 1 | |||||
| Li | [He] 2due south 1 | F | [He] 2s two 2p 5 | |||
| Na | [Ne] iiis 1 | Cl | [Ne] 3s 2 threep five | |||
| G | [Ar] 4due south 1 | Br | [Ar] 4southward two 3d 10 4p 5 | |||
| Rb | [Kr] fivesouth 1 | I | [Kr] vs 2 ivd 10 5p v | |||
| Cs | [Xe] 6s ane | At | [Xe] 6s 2 4f 14 vd ten sixp 5 |
The figure below shows the relationship between the periodic table and the orbitals being filled during the aufbau process. The two columns on the left side of the periodic tabular array stand for to the filling of an due south orbital. The next x columns include elements in which the five orbitals in a d subshell are filled. The six columns on the right represent the filling of the three orbitals in a p subshell. Finally, the 14 columns at the bottom of the tabular array correspond to the filling of the seven orbitals in an f subshell.

![]()
Allowed Set Of Quantum Numbers,
Source: https://chemed.chem.purdue.edu/genchem/topicreview/bp/ch6/quantum.html#:~:text=Rules%20Governing%20the%20Allowed%20Combinations%20of%20Quantum%20Numbers&text=The%20principal%20quantum%20number%20(n)%20cannot%20be%20zero.,between%200%20and%20n%20%2D%201.
Posted by: loefflergivy1941.blogspot.com


0 Response to "Allowed Set Of Quantum Numbers"
Post a Comment